Company Overview
NanoViricides, Inc. is a global leader in the development of nanomedicine drugs against viruses. Our unique nanoviricide® platform technology defines a novel mechanism enabling first-in class drugs against viruses.
A nanoviricide is designed to specifically attack enveloped virus particles, on the same sites that they use to bind to cells, and dismantle them, blocking reinfection cycle, going beyond what antibodies and immunotherapeutics do. A nanoviricide can also be designed to deliver anti-viral payload into infected cells sparing uninfected cells to block replication cycle without toxicity. Our unique biomimetic approach enables creation of drugs that a virus would be highly unlikely to escape due to mutations.
NanoViricides is one of the few biopharma companies with its own multi-kilogram-scale c-GMP capable manufacturing facility for the drug substance as well as drug product, in Shelton, CT. This facility is fully owned by the Company with no mortgage and is a major asset. This flexible, multi-product pilot plant can supply drug product for all of our programs through human clinical trials. Our cGMP manufacturing ability enables substantial time and cost savings in our drug development programs. Further, it is capable of production for initial marketing, enabling early revenues upon drug approval, of about $100MM~$500MM per year.
This first drug candidate is a topical treatment for shingles rash. Topical treatment has the advantage of delivering very high concentrations of the active ingredient (API) at the site of infection to eradicate the virus. We are already working on expansion to several additional indications in the HerpeCide™ program with variations of the same broad-spectrum drug, maximizing return on investments. These indications in the pipeline include: (ii) HSV-1 "cold sores", (iii) HSV-2 "genital ulcers", (iv) eye drops for the treatment of herpes keratitis (HK), (v) intravitreal injection to treat intra-ocular herpes and viral acute retinal necrosis (v-ARN; a cause of corneal transplants & blindness), among others. The market size for our HerpeCide program is in excess of ~$10Bn, with the market size for NV-HHV-101 estimated at between $0.5Bn~$2Bn by independent market studies.
In addition, the Company has drug candidates in development against influenzas (including bird flu), HIV, Dengue, Ebola/Marburg and other viruses at different preclinical stages. We have already proven animal model effectiveness and preliminary safety in many of these programs which are ready for candidate optimization and clinical candidate selection studies. These programs comprise a market size of $40~70 Billion.
This broad pipeline is enabled by our unique post-immunotherapeutic "bind-encapsulate-destroy" technology platform.
Unique, Novel, Post-ImmunoTherapeutic "Bind-Encapsulate-Destroy" Mechanism
Beyond Antibodies and Vaccines: Antibodies have been developed as drugs against viruses. However, each antibody only binds by two points to the virus, and destruction of the complex requires effective immune function, which is not the case in sickness. Vaccines only train the body into producing antibodies against the virus in the vaccine. Antibodies and vaccines are easily overcome by viruses by mutating in the field, hence the need for annual influenza vaccine updates.
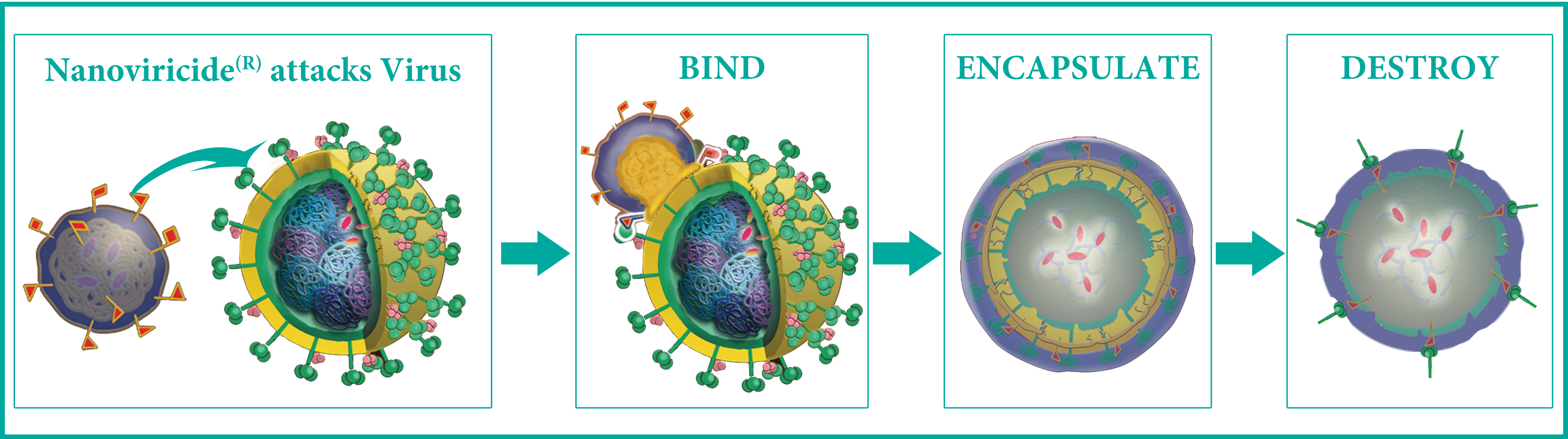
A Nanoviricide is A Nanomachine that Completes the Task of Destroying Virus without Help from the Patient's Immune System Key Issue of Drug Resistance from Viral Mutations is Unlikely with Nanoviricides Drugs due to Unique Biomimetic Technology
In contrast, a nanoviricide should work against all strains of the virus because they still bind the same way to the same host cell receptor. The nanoviricide® technology platform mimics the host cell. We design and develop a virus-binding ligand that mimics the site on the host cell receptor to which the virus binds. This ligand is then chemically attached to a special polymer to make a nanoviricide®. The virus is expected to be fooled into binding to the nanoviricide, like a venus-fly-trap. The nanoviricide is then expected to engulf the virus and possibly destroy it. A nanoviricide attacks the virus with hundreds of virus-binding sites on its surface. The nanoviricide is capable of dismantling the virus, and the resulting complexes are fully biodegradable in the body.
Strong Nanomedicine Intellectual Property Portfolio
NanoViricides, Inc. product candidates are based on TheraCour® technology invented and developed by company president and founder Anil R. Diwan, PhD. NanoViricides, Inc. holds an exclusive, worldwide license to this technology for its antiviral drugs. The technology is protected by two very broad international patent applications that have resulted in several international patents. These patents cover compositions of matter, processes of manufacture, methods of use, and fields of use. Additional patent applications are expected, and the Company intends to patent each drug separately as well.
Independent Researchers Perform Biological Testing
The Company has several collaborations at leading academic , private and federal laboratories. These independent researchers perform nanoviricides testing against various viral targets. The core intellectual property studies are performed by us. This drug development strategy saves on capital needs and provides unbiased data on our drug candidates.
Advancing Rapidly Towards FDA Submissions
The Company is focused on bringing its first drug candidate in the HerpeCide™ program into human clinical trials soon. This drug candidate, NV-HHV-101, was found to be well tolerated in both on-going GLP Safety/Toxicology studies as well as the completed non-GLP portion of the studies. We are awaiting reports from IND-enabling studies in progress for the preparation of an IND application. We have received a favorable response from the US FDA to our pre-IND application for NV-HHV-101.
Contact Information
NanoViricides, Inc.
1 Controls Drive,
Shelton, CT 06484
Phone: +1-203-937-6137
Phone: +1-888-591-3579
Fax: +1-203-859-5095
E-mail: info@nanoviricides.com
